Introduction
Diffuse Large B-Cell Lymphoma (DLBCL) is an aggressive form of non-Hodgkin lymphoma that most commonly affects older adults. While ~80% of patients achieve complete response to frontline therapy, roughly half are cured while the other half relapse and receive salvage treatment such as transplant or CAR-T therapy, either of which may be curative. Significant efficacy in later lines has been demonstrated with the recently approved bispecific therapies.
Due to the aggressiveness of DLBCL and currently available therapies that offer a potential cure, there is growing interest in a monitoring tool that provides additional information to standard disease monitoring where clinical imaging may fall short. clonoSEQ Ò is a next-generation sequencing CLIA LDT which monitors measurable residual disease (MRD) levels from ctDNA of DLBCL patients
Methods
The clonoSEQ assay sequences the V(D)J regions (specifically, the CDR3) of Ig loci. A baseline (calibration) sample containing high tumor burden is sequenced to determine the clonotypic/dominant sequence(s). The clonoSEQ algorithm assigns a uniqueness score to each dominant sequence based on analyses of Ig locus V, D, and J segments and non-templated nucleotides. This score reflects the probability a that sequence could be independently recreated in a non-malignant cell, including in another person, and is incorporated into the limit of detection (LOD). Consequently, sequences with a lower uniqueness must be observed at a higher rate to be above the LOD and reflect confidence they are tumor related in subsequent MRD samples.
Previous reports have explored the uniqueness distribution of Ig loci, but did not consider the impact of LOD on report interpretation. Using data from our database, we assessed ID sample calibration rates and Ig loci uniqueness among patients with orders from the clinical offering and from clinical trials. Our database contains 3657 dominant sequences from 1268 patients. 969 of these patients are from 27 clinical studies, and 302 are from use of the commercially available CLIA LDT assay.
Results:
Overall, 1535 ID samples have been received and included several different sample tissues and preparation methods. 1285 (83.7%) of these calibrated. Calibration was most successful in FFPE and gDNA preparations from lymphatic tissue or extranodal masses with 806/883 (91.3%). Least successful sample types included whole blood (48/106 [45.3%] calibrated) and skin (9/28 [32.1%] calibrated) which is likely a reflection of disease burden not being present in these tissues among a large proportion of the patients tested. For samples run on the clinical offering where the sample requirements are more stringent, the most common and most successfully calibrated sample type is FFPE lymph node tissue with a calibration rate of 95.2%.
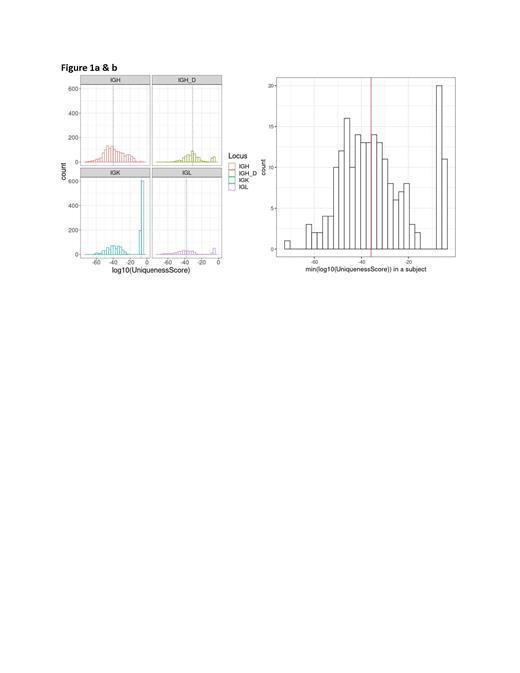
Across the population of 1268 DLBCL patients and 3657 dominant sequences, there was a range of 1-9 dominant sequences (avg 2.89; med 3). We assessed the distribution of uniqueness across the Ig loci in these patients. Fig 1a reflects a higher median uniqueness of IgH (VJ), IgH (DJ), and IgL clonotypes relative to IgK.
Fig 1b indicates the uniqueness score for each patient with only one dominant sequence, with just 31 (2.4%) of all subjects with expected ‘backgrounds’ of > 1/100,000 of all Ig sequences.
Conclusions
Our analysis of >1200 DLBCL patients demonstrates that identification of dominant Ig sequences is more successful using lymph tissue and that the majority of these patients rely on multiple dominant sequences for disease tracking. In a small subset of patients (2.4%), disease is only trackable using one lower uniqueness sequence. In these cases, the LOD is particularily informative in providing guidance to contextualize the probability the sequence may not be associated with the disease being monitoried.
Disclosures
Simmons:Adaptive Biotechnologies: Current Employment, Current equity holder in publicly-traded company. Lee:Adaptive Biotechnologies: Current Employment, Current equity holder in publicly-traded company. Lo:Adaptive Biotechnologies: Current Employment, Current equity holder in publicly-traded company. Akers:Adaptive Biotechnologies: Current Employment, Current equity holder in publicly-traded company. Kirsch:Adaptive Biotechnologies: Current Employment, Current equity holder in publicly-traded company. Jacob:Adaptive Biotechnologies: Current Employment, Current equity holder in publicly-traded company.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal